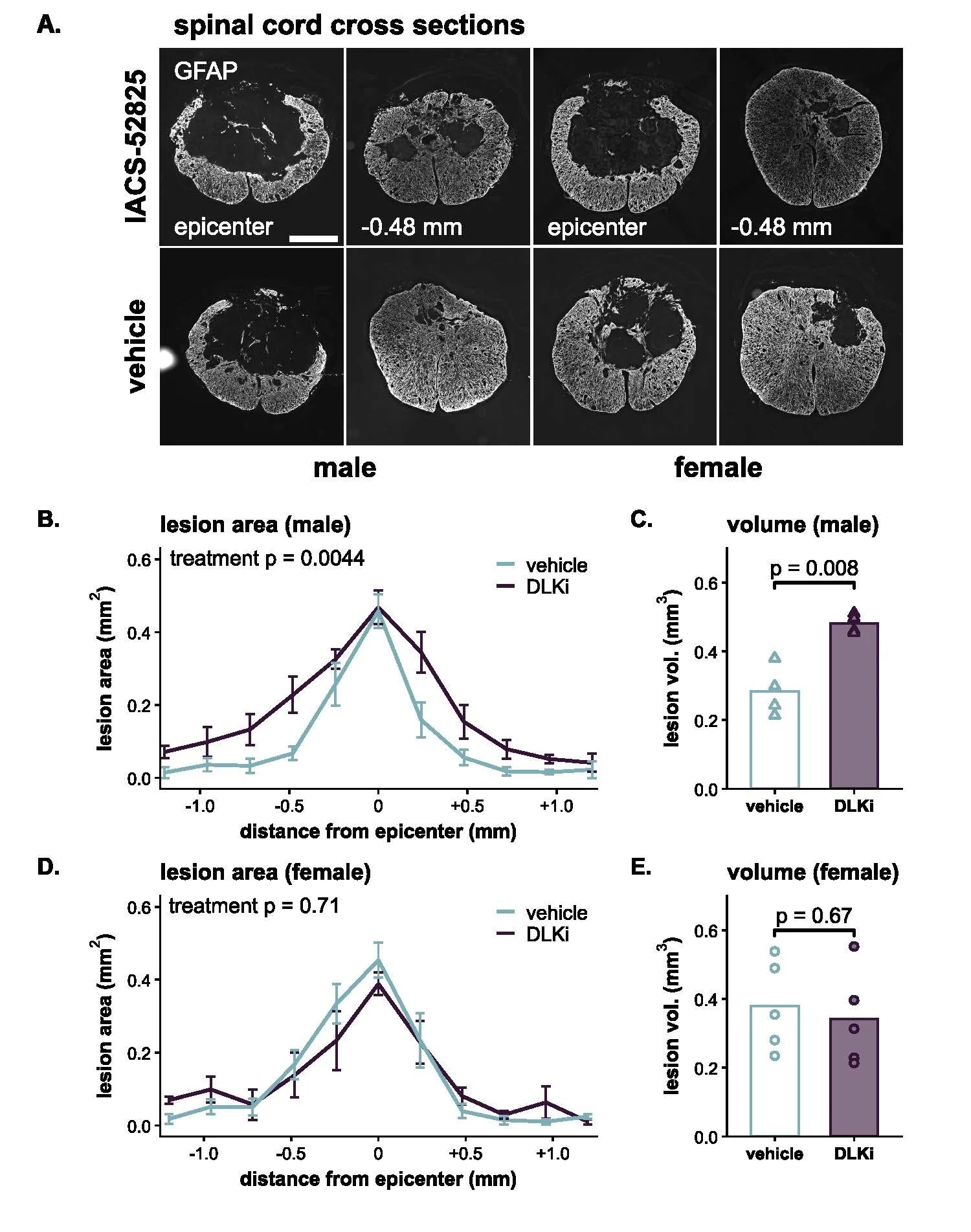
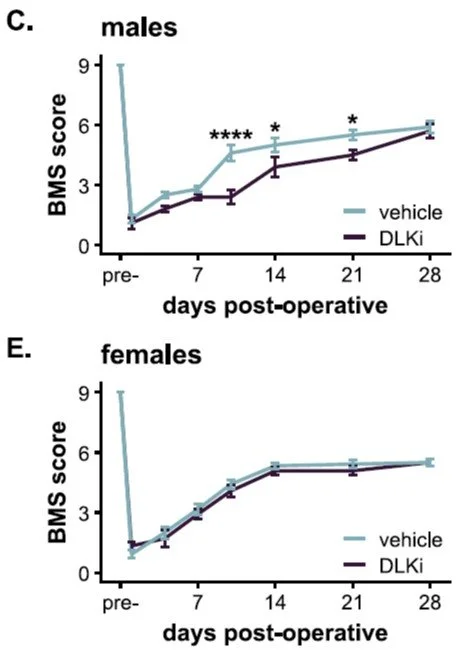
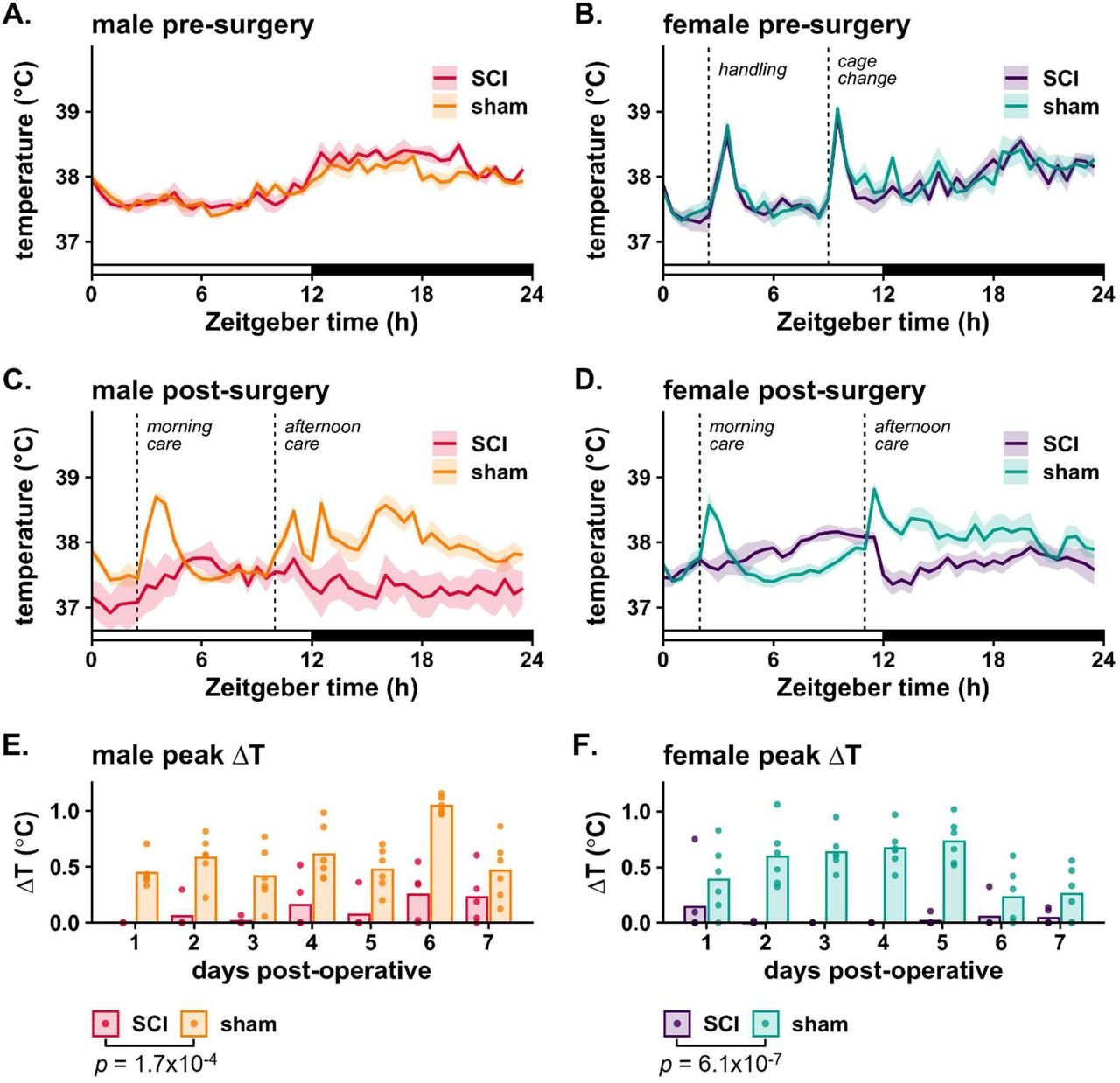
We are excited to share that our new review paper is featured on the cover of the March issue of Trends in Neurosciences!
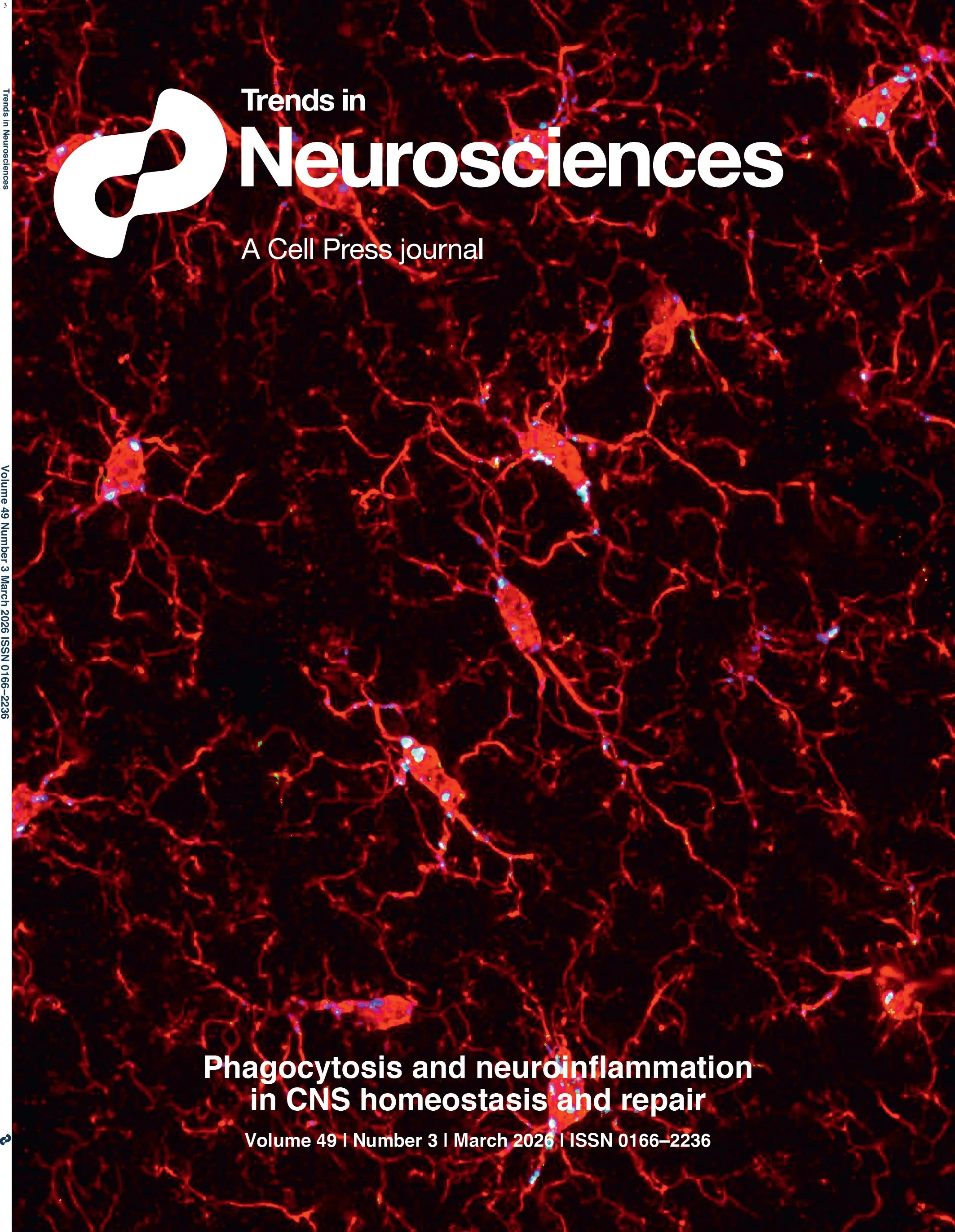
Trends in Neurosciences cover, March 2026 - Gaudet and Fonken. The cover shows hippocampal microglia (red, Iba1) with engulfed postsynaptic material (green, PSD95) in phagolysosomes (blue, CD68) during development. Image credit: Brandy Routh, Fonken lab.
The paper, “Phagocytosis and neuroinflammation: orchestrating CNS homeostasis, repair, and the resolution of inflammation,” was a collaboration with Laura Fonken (UT Austin College of Pharmacy). We developed a framework to describe how phagocytosis - the cellular ingestion of debris - crucially regulates the inflammatory response in the brain and spinal cord.
Beyond Cleanup: Phagocytosis as an Inflammatory Conductor
Phagocytosis is often oversimplified as a passive cleanup process. However, the molecules that drive debris clearance also dictate the inflammatory tone of the CNS. In our review, we explore how this double-edged sword impacts various pathologies:
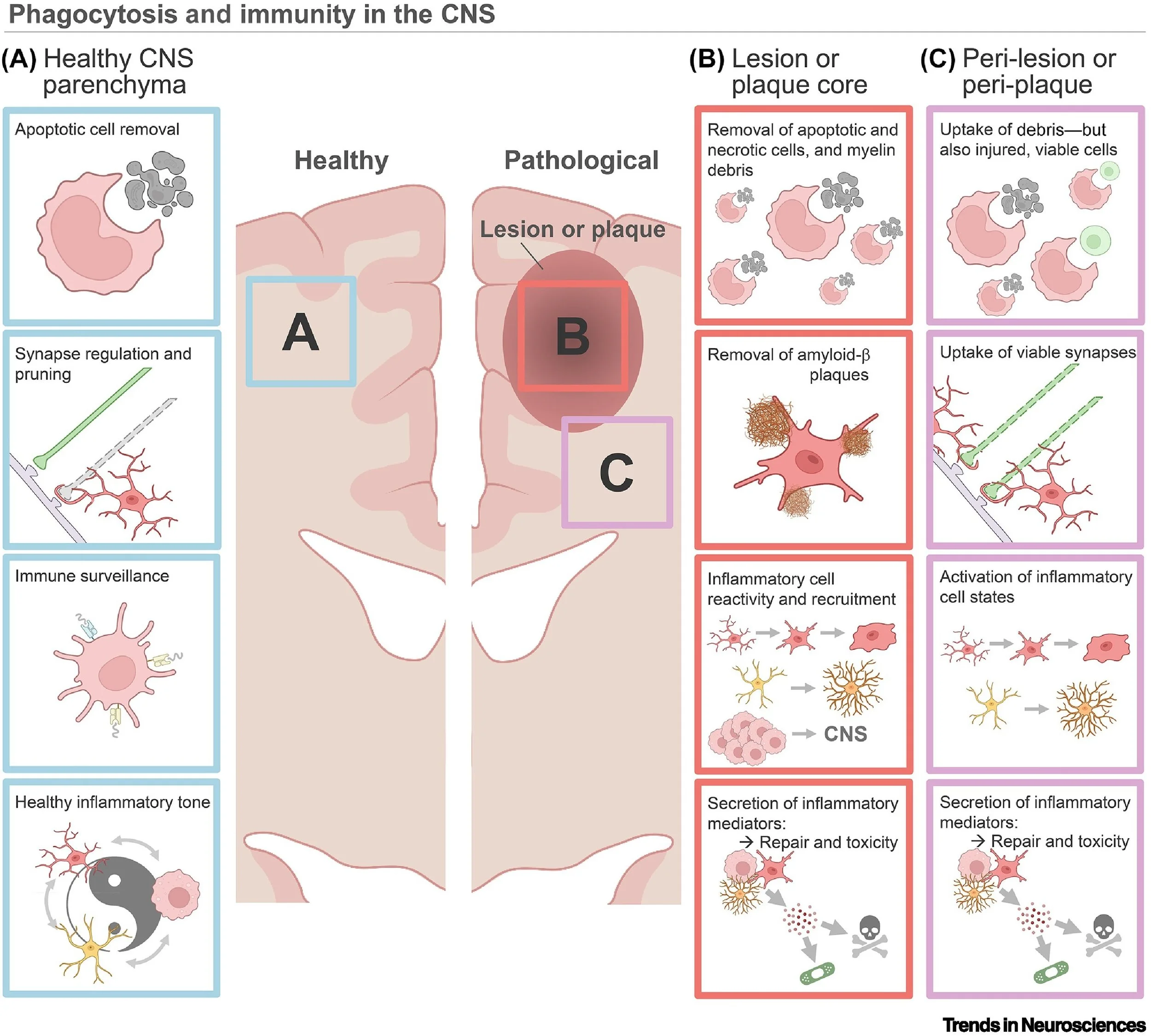
Homeostasis: Microglia and astrocytes manage synapse pruning and apoptotic cell removal to maintain health.
Disease and injury: The role of receptors like TREM2 and TAM receptors in clearing myelin debris in MS, amyloid-β in Alzheimer’s, and necrotic tissue after CNS injury and stroke.
Regulating debris removal and inflammatory state: We discuss how activating phagocytic systems can drive removal of damaging debris, while also shaping pro- versus anti-inflammatory cell state. These cell states have implications for disease and repair trajectories.
Complexities with "eat-me" signals: We highlight the delicate balance in the peri-lesion environment, where phagocytes must distinguish between toxic debris and viable, but stressed, neurons.
Figure 1: Phagocytosis and immunity in the central nervous system.
Future Therapeutic Directions
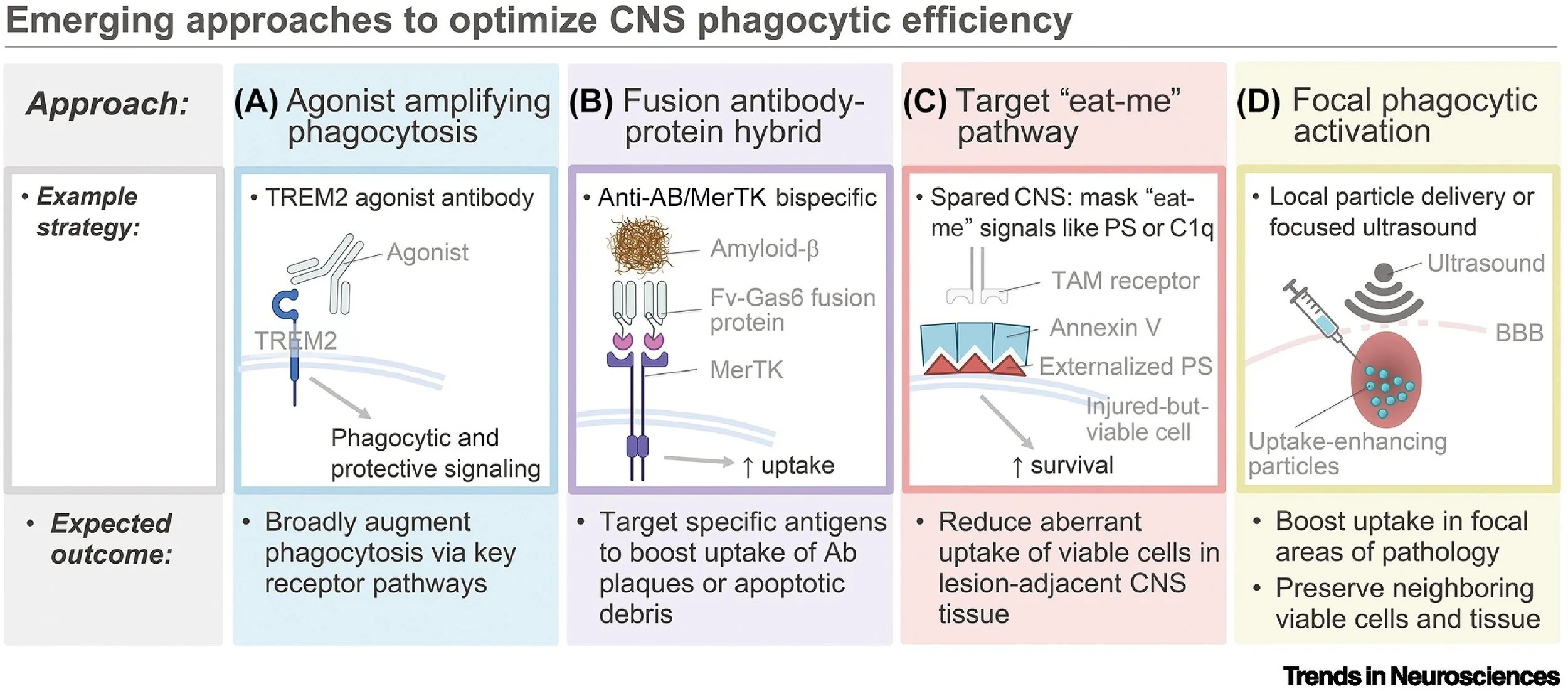
A major focus of the review is the transition from understanding mechanisms to fine-tuning these pathways to enhance recovery. We discuss emerging strategies - including TREM2 agonists, bispecific antibodies, and focused ultrasound - designed to optimize debris clearance while protecting healthy tissue.
Figure 3 from the paper: Emerging approaches to optimize CNS phagocytic efficiency.
We are grateful to the editors at Trends in Neurosciences for the cover feature and the opportunity to synthesize these evolving concepts. We hope this framework helps guide future research into these phagocytic regulators that resolve neuroinflammation and promote repair.
Check out the full study here: https://authors.elsevier.com/a/1mi1vbotqBbQu (temporary free link)
DOI: https://doi.org/10.1016/j.tins.2026.01.002
Authors: Andrew D. Gaudet and Laura K. Fonken